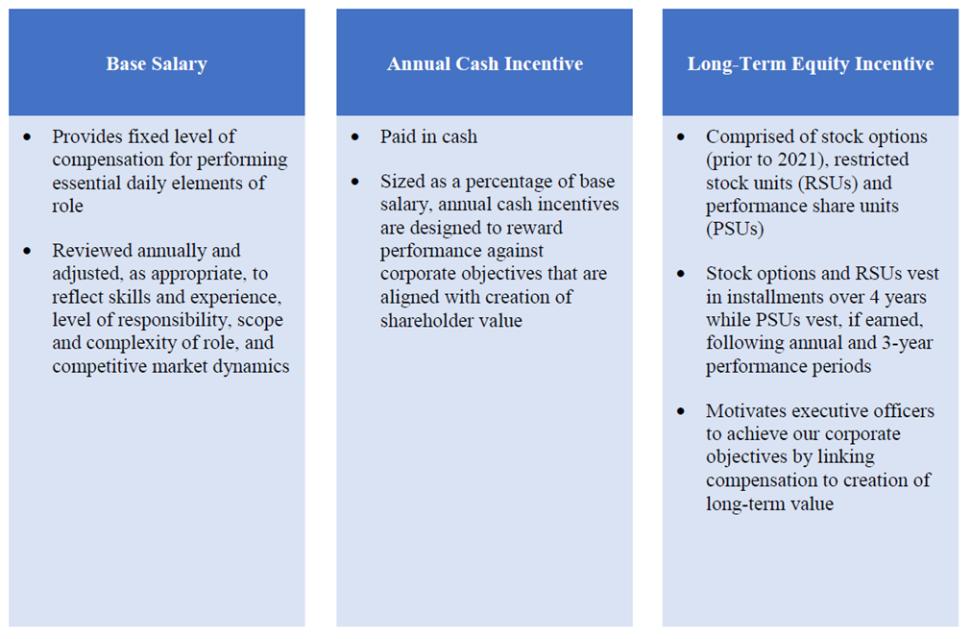
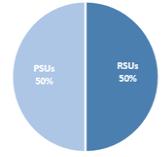
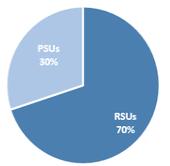
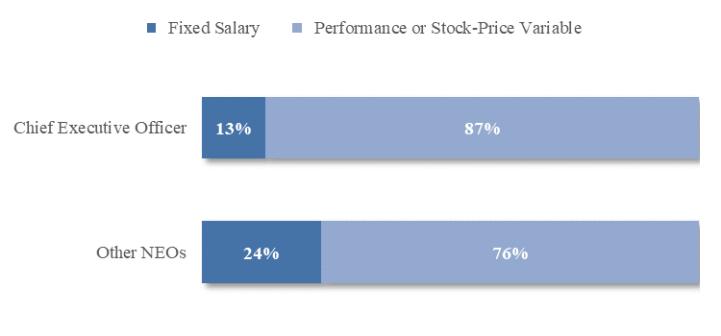
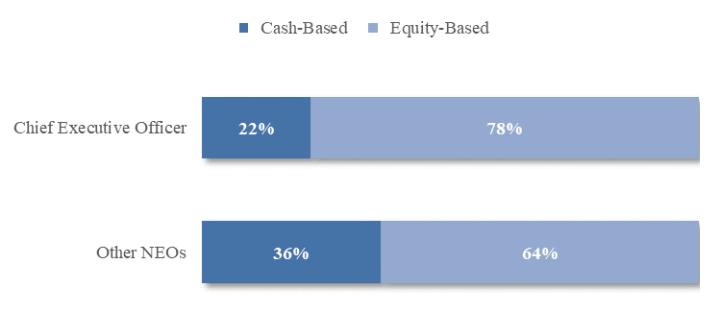
Option Exercises and Stock Vested
The following table sets forth certain information regarding option exercises and stock vested during the year ended December 31, 2020 with respect to us, an affiliated company or a business unit and are limited to one or more of the following: (i) specified levels of or increases in pre-tax earnings, return on capital, equity measures/ratios (on a gross, net, pre-tax or post tax basis), including basic earnings per share, diluted earnings per share, total earnings (including total earnings as adjusted by the compensation committee at the time of the Award), operating earnings, earnings growth, earnings before interest and taxes ("EBIT"), and earnings before interest, taxes, depreciation and amortization ("EBITDA") (including EBIT or EBITDA as adjusted by the committee at the time of the Award); (ii) total sales or sales growth; (iii) gross margin; (iv) customer service levels; (v) employee recruiting and development; (vi) advertising effectiveness; (vii) development of new markets; (viii) financial ratios; (ix) strategic initiatives; (x) improvement in or attainment of operating expense levels; (xi) improvement in or
Table of Contents
attainment of capital expense levels; (xii) the attainment of certain target levels of, or a specified increase in, operational cash flow; (xiii) the achievement of a certain level of, reduction of, or other specified objectives with regard to limiting the level of increase in, all or a portioneach of our bank debt or other long-term or short-term public or private debt or other similar financial obligations of ours, which may be calculated net of such cash balances and/or other specified offsets; (xiv) appreciation in and/or maintenance of certain target levels in the fair market value; (xv) the attainment of a certain level of, reduction of, or other specified objectives with regard to limiting the level of or rate of increase in all or a portion of specified expenses (xvi) individual objectives; and (xvii) any combination of the foregoing.
The compensation committee may impose restrictions on the grant, exercise or payment of an Award as it determines appropriate. Generally, Awards granted under the Plan will be nontransferable except by will or by the laws of descent and distribution. No participant will have any rights as a shareholder with respect to shares covered by options or RSUs, unless and until such Awards are settled in shares of common stock.
No option will be exercisable, no shares of common stock will be issued, no certificates for shares of common stock will be delivered and no payment will be made under the Plan except in compliance with all applicable laws.
�� The Board may amend, suspend or terminate the Plan and the compensation committee may amend any outstanding Award at any time; provided, however, that no such amendment or termination may adversely affect Awards then outstanding without the holder's permission.
In the event of a change in control (as defined in the Plan), the compensation committee may, on a participant-by-participant basis: (i) cause any or all outstanding Awards to become vested and immediately exercisable (as applicable), in whole or in part; (ii) cause any outstanding option to become fully vested and immediately exercisable for a reasonable period in advance of the change in control and, to the extent not exercised prior to that change in control, cancel that option upon closing of the change in control; (iii) cancel any unvested Award or unvested portion thereof, with or without consideration; (iv) cancel any Award in exchange for a substitute award; (v) redeem any restricted stock or RSU for cash and/or other substitute consideration with value equal to fair market value of an unrestricted share on the date of the change in control; (vi) cancel any outstanding options with respect to all common stock for which the Award remains unexercised for a cash payment equal to the excess (if any) of the fair market value of the shares subject to the option over the exercise price of the option; (vii) take such other action as the compensation committee will determine to be reasonable under the circumstances; and/or (viii) in the case of any Award subject to Section 409A of the Internal Revenue Code (the "Code"), such Award will vest and be distributed only in accordance with the terms of the applicable award agreement and the compensation committee will only be permitted to use discretion to the extent that such discretion would be permitted under Section 409A of the Code.
Neither the Board nor the compensation committee may, without obtaining prior approval of our shareholders: (i) implement any cancellation/re-grant program pursuant to which outstanding options under the Plan are cancelled and new options are granted in replacement with a lower exercise per share, (ii) cancel outstanding options under the Plan with an exercise price per share in excess of the then current fair market value per share for consideration payable in our equity securities or (iii) otherwise directly reduce the exercise price in effect for outstanding options under the Plan.
2015 Employee Stock Purchase Plan
On April 23, 2015, we adopted the 2015 Employee Stock Purchase Plan (the "2015 ESPP"), which became effective immediately prior to the closing of the IPO. The 2015 ESPP is administered by our Board or by a committee appointed by our Board. The 2015 ESPP provides participating employees with the opportunity to purchase an aggregate of 407,393 shares of our common stock, which includes
Table of Contents
shares of common stock that were automatically added to the shares reserved for issuance under the 2015 ESPP pursuant to an "evergreen" provision contained in the 2015 ESPP. Pursuant to such provision, the number of shares of our common stock reserved for issuance under the 2015 ESPP automatically increases on the first day of each fiscal year, commencing on January 1, 2016 and ending on December 31, 2025, in an amount equal to the least of (i) 400,000 shares of our common stock, (ii) 1.0% of the total number of shares of our common stock outstanding on the first day of the applicable year, and (iii) an amount determined by our Board.
All of our employees are eligible to participate in the 2015 ESPP, provided that:
•such person is customarily employed by us for more than 20 hours a week and for more than five months in a calendar year;
•such person has been employed by us or by a designated subsidiary for at least 21 days prior to enrolling in the 2015 ESPP; and
•such person was our employee on the first day of the applicable offering period under the 2015 ESPP.
No employee may purchase shares of our common stock under the 2015 ESPP and any of our other employee stock purchase plans in excess of $25,000 of the fair market value of our common stock (as of the date of the option grant) in any calendar year. In addition, no employee may purchase shares of our common stock under the 2015 ESPP that would result in the employee owning 5% or more of the total combined voting power or value of our stock.
We expect to make one or more offerings to our eligible employees to purchase stock under the 2015 ESPP beginning at such time as our Board (or a person designated by our Board) may determine. Each offering will consist of a six-month offering period during which payroll deductions will be made and held for the purchase of our common stock at the end of the offering period. The first such offering period began on February 1, 2016 and will end on July 30, 2016. Our Board may, at its discretion, choose a different length of offering period so long as it does not exceed 12 months.
On the commencement date of each offering period, each eligible employee may authorize up to a maximum of 15% of his or her compensation to be deducted by us during the offering period. Each employee who continues to be a participant in the 2015 ESPP on the last business day of the offering period will be deemed to have exercised an option to purchase from us the number of whole shares of our common stock that his or her accumulated payroll deductions on such date will pay for, not in excess of the maximum numbers set forth above. Under the terms of the 2015 ESPP, the purchase price will be determined by our Board or compensation committee for each offering period and will be at least 85% of the applicable closing price of our common stock. If our Board does not make a determination of the purchase price, the purchase price will be 85% of the lesser of the closing price of our common stock on the first business day of the offering period or on the last business day of the offering period.
An employee may for any reason withdraw from participation in an offering no later than 21 days prior to the end of an offering period and permanently draw out the balance accumulated in the employee's account. If an employee elects to discontinue his or her payroll deductions during an offering period but does not elect to withdraw his or her funds, funds previously deducted will be applied to the purchase of common stock at the end of the offering period. If a participating employee's employment ends before the last business day of an offering period, no additional payroll deductions will be made and the balance in the employee's account will be paid to the employee.
We will be required to make equitable adjustments to the number and class of securities available under the 2015 ESPP, the share limitations under the 2015 ESPP and the purchase price for an offering period under the 2015 ESPP to reflect stock splits, reverse stock splits, stock dividends,
Table of Contents
recapitalizations, combinations of shares, reclassifications of shares, spin-offs and other similar changes in capitalization or events or any dividends or distributions to holders of our common stock other than ordinary cash dividends. The number of shares of common stock available for any offerings under the 2015 ESPP will be adjusted if the number of our outstanding shares is increased or reduced by split up, reclassification, stock dividend or the like.
Our Board may at any time, and from time to time, terminate, amend or suspend the 2015 ESPP or any portion thereof. We will obtain shareholder approval for any amendment if such approval is required by Section 423 of the Code. The 2015 ESPP will also terminate upon the occurrence of a change in control (defined with reference to the Plan). Upon termination, we will refund all amounts in the accounts of participating employees.
Non-Equity Incentive Compensation
Performance Bonus Plan
On April 23, 2015, we adopted the Performance Bonus Plan, which became effective immediately prior to the closing of the IPO. The Performance Bonus Plan is administered by the compensation committee. The purpose of the Performance Bonus Plan is to benefit and advance our interests, by rewarding selected employees of ours and our affiliates for their contributions to our success and thereby motivate them to continue to make such contributions in the future by granting performance-based awards. The material terms of such plan are summarized below.
Background. Our Board believes that it is in our best interests and those of our shareholders to enhance our ability to attract and retain qualified personnel through performance based incentives, and once we are subject to Section 162(m) of the Code, maintain the highest level of deductibility of compensation paid to employees. Section 162(m) provides newly public companies an opportunity to transition their compensation structures to comply with the rules under Section 162(m), and we will generally become subject to such rules beginning with our 2019 annual meeting of shareholders. Section 162(m) of the Code disallows a deduction to us for any compensation paid to certain executive officers in excess of $1.0 million per year, subject to certain exceptions. Among other exceptions, the deduction limit does not apply to compensation that meets the specified requirements for "performance-based compensation." In general, those requirements include the establishment of objective performance goals for the payment of such compensation by a committee of the Board composed solely of two or more outside directors, shareholder approval of the material terms of such compensation, and certification by the compensation committee that the performance goals for the payment of such compensation have been achieved.
Administration. Subject to the other provisions of the Performance Bonus Plan, the compensation committee has the authority to administer and interpret the Performance Bonus Plan, including the authority to select the employees (including employees who are directors) to participate in the Performance Bonus Plan, to establish the performance goals, to determine the amount of incentive compensation bonus payable to any participant, to determine the terms and conditions of any such incentive opportunity; to make all determinations and take all other actions necessary or appropriate for proper administration and operation of the Performance Bonus Plan and to establish and amend rules and regulations relating to the Performance Bonus Plan.
The compensation committee may also delegate to one or more of our executive officers the authority to administer the Performance Bonus Plan with respect to any participants who are not subject to Section 162(m) of the Code.
Eligibility. The named executive officers and such other of our employees as selected by the compensation committee are eligible to participate in the Performance Bonus Plan. The maximum amount of the incentive compensation bonuses payable to any participant under the Performanceofficers:
| | | | | |
| Option Awards | | Stock Awards |
| Number of Shares
Acquired on Exercise | Value Realized on
Exercise | | Number of Shares
Acquired on Vesting | Value Realized on
Vesting |
Name | (#) | ($) | | (#) | ($) |
Joseph Ciaffoni | 64,820 | 862,994 | | 56,102 | 1,150,798 |
President and Chief Executive Officer | | | | | |
| | | | | |
Paul Brannelly | - | - | | 18,927 | 395,089 |
Executive Vice President and Chief Financial Officer | | | | | |
| | | | | |
Alison Fleming, Ph.D. | 16,000 | 218,400 | | 17,327 | 360,497 |
Executive Vice President and Chief Technical Officer | | | | | |
| | | | | |
Shirley Kuhlmann | - | - | | 11,400 | 225,218 |
Executive Vice President and General Counsel | | | | | |
| | | | | |
Richard Malamut, M.D. | - | - | | 10,625 | 164,581 |
Executive Vice President and Chief Medical Officer | | | | | |
Table of Contents
Bonus Plan in, or in respect of, any single fiscal year will not exceed $5.0 million. All incentive compensation bonuses paid pursuant to the Performance Bonus Plan will be paid in cash.
Bonus Opportunity and Performance Goals. Bonuses may be payable to a participant as a result of the satisfaction of performance goals in respect of any performance period determined by the compensation committee; provided that, to the extent a participant would be subject to Section 162(m) of the Code, the performance goals will be set in accordance with the regulations under Section 162(m) of the Code. Performance goals, which may vary among and between participants, may include objectives stated with respect to us, an affiliated company or a business unit and such objectives are limited to one or more of the following: (i) specified levels of or increases in pre-tax earnings, return on capital, equity measures/ratios (on a gross, net, pre-tax or post tax basis), including basic earnings per share, diluted earnings per share, total earnings (including total earnings as adjusted by the compensation committee at the time of the Award), operating earnings, earnings growth, EBIT and EBITDA (including EBIT or EBITDA as adjusted by the compensation committee at the time of the Award); (ii) total sales or sales growth; (iii) gross margin; (iv) customer service levels; (v) employee recruiting and development; (vi) advertising effectiveness; (vii) development of new markets; (viii) financial ratios; (ix) strategic initiatives; (x) improvement in or attainment of operating expense levels; (xi) improvement in or attainment of capital expense levels; (xii) the attainment of certain target levels of, or a specified increase in, operational cash flow; (xiii) the achievement of a certain level of, reduction of, or other specified objectives with regard to limiting the level of increase in, all or a portion of, the Company's bank debt or other long-term or short-term public or private debt or other similar financial obligations of the Company, which may be calculated net of such cash balances and/or other specified offsets; (xiv) appreciation in and/or maintenance of certain target levels in the fair market value; (xv) the attainment of a certain level of, reduction of, or other specified objectives with regard to limiting the level of or rate of increase in all or a portion of specified expenses (xvi) individual objectives; and (xvii) any combination of the foregoing.
The compensation committee will provide a threshold level of performance below which no incentive compensation bonus will be paid, as well as a maximum level of performance above which no additional incentive compensation bonus will be paid. It also may provide for the payment of differing amounts for different levels of performance, determined with regard either to a fixed monetary amount or a percentage of the participant's base salary. The compensation committee will make such adjustments, to the extent it deems appropriate, to established performance goals and performance thresholds to compensate for, or to reflect, any material changes which may have occurred due to an "extraordinary event"; provided, however, that no such adjustment may be made unless such adjustment would be permissible under Section 162(m) of the Code. An "extraordinary event" under the Performance Bonus Plan is defined as follows:
•material changes in accounting practices, tax laws, other laws or regulations,
•material changes in our financial structure,
•an acquisition or disposition of one of our subsidiaries or divisions, or
•unusual circumstances outside of our management's control which, in the sole judgment of the compensation committee, alters or affects the computation of such established performance goals and performance thresholds, our performance or the performance of a relevant subsidiary or division.
As soon as practicable after the end of each performance period, but before any incentive compensation bonuses are paid to the participants under the Performance Bonus Plan, the compensation committee will certify in writing (i) whether the performance goal(s) were attained and (ii) the amount of the incentive compensation bonus payable to each participant based upon the attainment of such specified performance goals. The compensation committee also may reduce,
Table of Contents
eliminate, or, with respect only to participants who are not subject to Section 162(m) of the Code, increase the amount of any incentive compensation bonus of any participant at any time prior to payment thereof, based on such criteria as the compensation committee will determine, including but not limited to individual merit and attainment of, or the failure to attain, specified personal goals established by the compensation committee. Under no circumstances, however, may the compensation committee, with respect solely to a participant who is subject to Section 162(m) of the Code, (i) increase the amount of the incentive compensation otherwise payable to such participant beyond the amount originally established by the compensation committee, (ii) waive the attainment of the performance goals established and applicable to such participant's incentive compensation or (iii) otherwise exercise its discretion so as to cause any incentive compensation bonus payable to such participant to not qualify as "performance-based compensation" under Section 162(m) of the Code.
All amounts due under the Performance Bonus Plan will be paid within two and one-half months of the end of the year in which such incentive compensation is no longer subject to a risk of forfeiture. The compensation committee, without the consent of any participant, may amend or terminate the Performance Bonus Plan at any time. However, no amendment that would require the consent of the shareholders pursuant to Section 162(m) of the Code will be effective without such consent.
Retirement Benefits
We maintain a 401(k) Plan for all employees after three months of consecutive employment who are 2118 years of age or older. Employees can contribute up to 100%90% of their eligible pay, subject to maximum amounts allowed under law. We currently provide matching and profit sharing contributions under the 401(k) Plan. The total amount of our matching contributions under the 401(k) Plan for 2014 and 2015 was $35,000 and $44,095, respectively.
Table of Contents
CERTAIN RELATIONSHIPS AND RELATED PARTY TRANSACTIONS
The following includes a summary of transactions since January 1, 20152020 to which we have been a party, in which the amount involved in the transaction exceeded $120,000, and in which any of our directors, executive officers or, to our knowledge, beneficial owners of more than 5% of our capital stock or any member of the immediate family of any of the foregoing persons had or will have a direct or indirect material interest, other than equity and other compensation, termination, change in control and other arrangements, which are described elsewhere in this proxy statement.
Participation in Follow-On Public Offering
In January 2016, RA Capital Management LLC, which was previously one of our 5% shareholders, purchased an aggregate of 200,000 shares of our common stock in a follow-on public offering of our common stock at the public offering price. In addition, Eventide Asset Management, LLC and Healthcor Management LP, which are currently among our 5% shareholders but were not at the time of the follow-on public offering, purchased 275,000 and 150,000 shares of our common stock, respectively.
Participation in Initial Public Offering
In the IPO, certain of our 5% shareholders and their affiliates purchased an aggregate of 1,300,000 shares of our common stock. Each of those purchases was made through the underwriters at the initial public offering price. The following table set forth the aggregate number of shares of our common stock that these 5% shareholders and their affiliates purchased in the IPO:
| | | | |
Purchaser
| | Shares of
Common Stock | |
|---|
Frazier Healthcare VI, L.P.(1)
| | | 150,000 | |
Entities affiliated with Longitude Capital Partners, LLC(2)
| | | 200,000 | |
Skyline Venture Partners V, L.P.(3)
| | | 150,000 | |
TPG Biotechnology Partners IV, L.P.(4)
| | | 100,000 | |
RA Capital Healthcare Fund, LP(5)
| | | 700,000 | |
Eventide Asset Management, LLC(6)
| | | 450,000 | |
(1)The general partner of Frazier Healthcare VI, L.P. is a limited partnership, the general partner of which is FHM VI, LLC. Patrick Heron, a member of our Board, is a member of FHM VI, LLC.
(2)Represents shares held, in the aggregate, by Longitude Venture Partners, L.P. and Longitude Capital Associates, L.P. or, collectively, the Longitude Funds. Longitude Capital Partners, LLC is the general partner of the Longitude Funds. David Hirsch, a member of our Board, is a member of Longitude Capital Partners, LLC.
(3)The general partner of Skyline Venture Partners V, L.P. is Skyline Venture Management V, LLC. John G. Freund, a member of our Board, is a manager of Skyline Venture Management V, LLC.
(4)The general partner of TPG Biotechnology Partners IV, L.P. is TPG Biotechnology GenPar IV, L.P., a Delaware limited partnership, whose general partner is TPG Biotechnology GenPar IV Advisors, LLC, a Delaware limited liability company, whose sole member is TPG Holdings I, L.P., a Delaware limited partnership, whose general partner is TPG Holdings I-A, LLC, a Delaware limited liability company, whose sole member is TPG Group Holdings (SBS), L.P., a Delaware limited partnership, whose general partner is TPG Group Holdings (SBS) Advisors, Inc., or Group Advisors, a
Table of Contents
Delaware corporation. Messrs. David Bonderman and James G. Coulter are officers and the sole shareholders of Group Advisors and may therefore be deemed to be the beneficial owners of the shares held by TPG Biotechnology Partners IV, L.P. Each of Messrs. Bonderman and Coulter expressly disclaims beneficial ownership of the securities listed above except to the extent of any pecuniary interest therein. Eran Nadav, Ph.D., a member of our Board, is a partner of TPG Global, LLC. The address for Messrs. Bonderman, Coulter, Nadav and TPG Biotechnology Partners IV, L.P. is c/o TPG Global, LLC, 301 Commerce Street, Suite 3300, Fort Worth, TX 76102.
(5)The investment adviser and sole general partner of RA Capital Healthcare Fund, LP is RA Capital Management, LLC. Peter Kolchinsky is the sole managing member of RA Capital Management, LLC and has the power to vote or dispose of the shares held by RA Capital Healthcare Fund, LP. The address for Dr. Kolchinsky and RA Capital Healthcare Fund, LP is 20 Park Plaza, Suite 1200, Boston, MA 02116.
(6)Eventide Asset Management, LLC is currently one of our 5% shareholders but was not at the time of the IPO.
Series D Convertible Preferred Stock Financing
In March 2015, we entered into a Series D Convertible Preferred Stock Purchase Agreement pursuant to which we issued and sold to investors an aggregate of 41,666,667 shares of our Series D Convertible Preferred Stock at a purchase price of $1.20 per share, for aggregate consideration of $50.0 million. This financing was led by TPG Biotechnology Partners IV, L.P., which was joined by RA Capital Management, Adage Capital Management, Rock Springs Capital, EcoR1 Capital, Eventide Asset Management and Aperture Venture Partners. Entities affiliated with Longitude Capital Partners, LLC, Skyline Ventures Partners V, L.P., Frazier Healthcare VI, L.P. and Boston Millennia Partners also participated in the financing. In connection with the Series D Convertible Preferred Stock financing, convertible notes with related parties in the aggregate principal amount of $5 million automatically converted to an aggregate of 4,166,667 shares of Series D Convertible Preferred Stock.
Series C Convertible Preferred Stock Financing
In August 2013, we entered into a Series C Convertible Preferred Stock Purchase Agreement, or the Series C Purchase Agreement, pursuant to which we issued and sold to investors an aggregate of 2,220,670 shares of our Series C Convertible Preferred Stock at a purchase price of $1.386 per share, for aggregate consideration of $3.1 million. In September 2013, at an additional closing pursuant to the Series C Purchase Agreement, we issued and sold to investors an aggregate of 665,334 shares of our Series C Convertible Preferred Stock at a purchase price of $1.386 per share, for aggregate consideration of $922,000. In December 2013, those investors who participated in the closings in August and September 2013 exercised their option under the Series C Purchase Agreement to purchase an additional pro rata portion of an aggregate of 5,772,004 shares of our Series C Convertible Preferred Stock at a purchase price of $1.386 per share, for aggregate consideration of $8.0 million.
Series B Convertible Preferred Stock Financing
In February 2012, we entered into a Series B Convertible Preferred Stock Purchase Agreement, pursuant to which we issued and sold to investors an aggregate of 27,324,237 shares of our Series B Convertible Preferred Stock at a purchase price of $0.84 per share, for aggregate consideration of $23.0 million. In connection with the Series B Convertible Preferred Stock financing, all issued and outstanding shares of previously issued Series A Preferred Stock, Series B Preferred Stock, Series C Preferred Stock, Series C-2 Preferred Stock and Series D-1 Preferred Stock were converted into 18,464,674 shares of Series A Convertible Preferred Stock.
Table of Contents
All outstanding convertible preferred stock converted into common stock upon the closing of the IPO.
The participants in the convertible preferred stock financings described above included the following directors, executive officers and/or holders of more than 5% of our capital stock or entities affiliated with them. The following table presents the number of shares issued to these related parties in these financings:
| | | | | | | | | | | | | | | | | | | | | | | | | |
Participants(1) | | Shares of
Series A
Convertible
Preferred
Stock | | Series A
Convertible
Preferred
Stock
Aggregate
Purchase
Price | | Shares of
Series B
Convertible
Preferred
Stock | | Series B
Convertible
Preferred
Stock
Aggregate
Purchase
Price | | Shares of
Series C
Convertible
Preferred
Stock | | Series C
Convertible
Preferred
Stock
Aggregate
Purchase
Price | | Shares of
Series D
Convertible
Preferred
Stock | | Series D
Convertible
Preferred
Stock
Aggregate
Purchase
Price | |
|---|
5% or greater shareholders | | | | | | | | | | | | | | | | | | | | | | | | | |
Entities affiliated with Boston Millennia Partners(2) | | | 4,881,801 | (8) | $ | 5,988,017 | | | 1,862,481 | | $ | 1,564,484 | | | — | | $ | — | | | 712,357 | | $ | 854,828 | |
Frazier Healthcare VI, L.P.(3) | | | 4,019,183 | (8) | $ | 4,929,930 | | | 1,533,399 | | $ | 1,288,055 | | | 2,705,585 | | $ | 3,749,941 | | | 3,676,078 | | $ | 4,411,294 | |
Entities affiliated with Longitude Capital Partners, LLC(4) | | | — | | $ | — | | | 13,095,238 | | $ | 11,000,000 | | | 3,224,261 | | $ | 4,468,826 | | | 5,146,509 | | $ | 6,175,811 | |
Skyline Venture Partners V, L.P.(5) | | | — | | $ | — | | | 10,714,286 | | $ | 9,000,000 | | | 2,638,030 | | $ | 3,656,310 | | | 5,881,724 | | $ | 7,058,068 | |
TPG Biotechnology Partners IV, L.P.(6) | | | — | | | — | | | — | | | — | | | — | | | — | | | 8,333,333 | | $ | 10,000,000 | |
RA Capital Healthcare Fund, LP(7) | | | — | | | — | | | — | | | — | | | — | | | — | | | 5,833,333 | | $ | 7,000,000 | |
(1)Additional details regarding these shareholders and their equity holdings are provided in "Security Ownership of Certain Beneficial Owners and Management."
(2)Represents shares held, in the aggregate, by Boston Millennia Partners II Limited Partnership, Boston Millennia Partners II-A Limited Partnership, Boston Millennia Partners GmbH and Co. KG ("BMP KG"), Boston Millennia Associates II Partnership, and Strategic Advisors Fund Limited Partnership (collectively, the "Boston Millennia Funds"). Robert Jevon, a former member of our Board, is an affiliate of Glen Partners II Limited Partnership, which is the special limited partner of BMP KG, and the general partner of the other Boston Millennia Funds.
(3)The general partner of Frazier Healthcare VI, L.P. is a limited partnership, the general partner of which is FHM VI, LLC. Patrick Heron, a member of our Board, is a member of FHM VI, LLC.
(4)Represents shares held, in the aggregate, by Longitude Venture Partners, L.P. and Longitude Capital Associates, L.P. or, collectively, the Longitude Funds. Longitude Capital Partners, LLC is the general partner of the Longitude Funds. David Hirsch, a member of our Board, is a member of Longitude Capital Partners, LLC.
(5)The general partner of Skyline Venture Partners V, L.P. is Skyline Venture Management V, LLC. John G. Freund, a member of our Board, is a manager of Skyline Venture Management V, LLC.
(6)The general partner of TPG Biotechnology Partners IV, L.P. is TPG Biotechnology GenPar IV, L.P., a Delaware limited partnership, whose general partner is TPG Biotechnology GenPar IV Advisors, LLC, a Delaware limited liability company, whose sole member is TPG Holdings I, L.P., a Delaware limited partnership, whose general partner is TPG Holdings I-A, LLC, a Delaware limited liability company, whose sole member is TPG Group Holdings (SBS), L.P., a Delaware limited partnership, whose general partner is TPG Group Holdings (SBS) Advisors, Inc., or Group Advisors, a Delaware corporation. Messrs. David Bonderman and James G. Coulter are officers and the sole shareholders of Group Advisors and may therefore be deemed to be the beneficial owners of the shares held by TPG Biotechnology Partners IV, L.P. Each of Messrs. Bonderman and Coulter expressly disclaims beneficial ownership of the securities listed above except to the extent of any pecuniary interest therein. Eran Nadav, Ph.D., a member of our Board, is a partner of TPG Global, LLC. The address for Messrs. Bonderman, Coulter, Nadav and TPG Biotechnology Partners IV, L.P. is c/o TPG Global, LLC, 301 Commerce Street, Suite 3300, Fort Worth, TX 76102.
(7)The investment adviser and sole general partner of RA Capital Healthcare Fund, LP is RA Capital Management, LLC. Peter Kolchinsky is the sole managing member of RA Capital Management, LLC and has the power to vote or dispose of the shares held by RA Capital Healthcare Fund, LP. The address for Dr. Kolchinsky and RA Capital Healthcare Fund, LP is 20 Park Plaza, Suite 1200, Boston, MA 02116
(8)These amounts reflect the December 2013 reverse stock split pursuant to which every two issued and outstanding shares of Series A Convertible Preferred Stock were reclassified and combined into one share of Series A Convertible Preferred Stock.
Convertible Note Financing
In November and December 2014, we entered into a Convertible Note Purchase Agreement, pursuant to which we issued and sold to investors convertible promissory notes in the aggregate principal amount of $5.0 million (the "2014 Convertible Note Financing"). Pursuant to the Convertible Note Purchase Agreement, the convertible notes bore interest at a rate per annum of 6.0% and mature on the earlier of (i) November 14, 2015, (ii) immediately prior to a liquidation of the company, or (iii) upon an event of default. In connection with the Series D Convertible Preferred Stock financing, the notes automatically converted into an aggregate of 4,166,667 shares of Series D Convertible
Table of Contents
Preferred Stock. The participants in the 2014 Convertible Notes Financing included the following directors, executive officers and/or holders of more than 5% of our capital stock or entities affiliated with them. The following table presents the amount of the notes issued to these related parties in this financing:
| | | | |
Participants(1) | | Amount of
Notes Purchased | |
|---|
5% or greater shareholders | | | | |
Entities affiliated with Boston Millennia Partners(2) | | $ | 754,829 | |
Frazier Healthcare VI, L.P.(3) | | $ | 924,264 | |
Entities affiliated with Longitude Capital Partners, LLC(4) | | $ | 1,826,499 | |
Skyline Venture Partners V, L.P.(5) | | $ | 1,494,408 | |
(1)Additional details regarding these shareholders and their equity holdings are provided in "Security Ownership of Certain Beneficial Owners and Management."
(2)Represents shares held, in the aggregate, by the Boston Millennia Funds. Robert Jevon, a former member of our Board, is an affiliate of Glen Partners II Limited Partnership, which is the special limited partner of BMP KG, and the general partner of the other Boston Millennia Funds.
(3)The general partner of Frazier Healthcare VI, L.P. is a limited partnership, the general partner of which is FHM VI, LLC. Patrick Heron, a member of our Board, is a member of FHM VI, LLC.
(4)Represents shares held, in the aggregate, by the Longitude Funds. Longitude Capital Partners, LLC is the general partner of the Longitude Funds. David Hirsch, a member of our Board, is a member of Longitude Capital Partners, LLC.
(5)The general partner of Skyline Venture Partners V, L.P. is Skyline Venture Management V, LLC. John G. Freund, a member of our Board, is a manager of Skyline Venture Management V, LLC.
In connection with the 2014 Convertible Note Financing, we entered into a Preferred Shareholder Agreement with the Boston Millennia Funds, the Longitude Funds, Skyline Venture Partners V, L.P. and Frazier Healthcare VI, L.P., pursuant to which the Longitude Funds, Skyline Venture Partners V, L.P. and Frazier Healthcare VI, L.P. agreed to vote their shares to waive, with respect to the Boston Millennia Funds, application of a mandatory conversion provision in our articles of incorporation and other similar conversion provisions or other devices or mechanisms that may be adopted that are intended to incentivize the continued investment in the Company by existing holders of our preferred stock, subject to certain conditions described in the Preferred Shareholder Agreement. The Preferred Shareholder Agreement terminated by its terms upon the closing of the Series D Convertible Preferred Stock financing.
Seventh Amended and Restated Stockholders Agreement
In connection with the Series D Convertible Preferred Stock financing in March 2015, we entered into the Seventh Amended and Restated Stockholders Agreement (the "Stockholders Agreement") with certain of our shareholders, including Island View Investors, LLC, a limited liability company of which our President and Chief Executive Officer, Michael T. Heffernan, is the sole member, the Longitude Funds, the Boston Millennia Funds, Frazier Healthcare VI, L.P., Skyline Venture Partners V, L.P., TPG Biotechnology Partners IV, L.P. and RA Capital Healthcare Fund, LP. The Stockholders Agreement, among other things:
•imposed restrictions on the transfer of capital stock;
Table of Contents
•granted holders of our outstanding convertible preferred stock certain rights of first refusal, co-sale and put rights with respect to certain proposed transfers of our securities by shareholders other than holders of Series D Convertible Preferred Stock;
•granted us certain rights of first refusal with respect to certain proposed transfers of our securities by shareholders other than holders of Series D Convertible Preferred Stock;
•imposed "drag along" obligations on our shareholders, and permits "tag along" by holders of our preferred stock in certain sale transactions that are approved by our Board and certain of our preferred shareholders;
•set forth voting obligations with respect to the constituency and size of our Board, and provides for the designation of our directors by certain of our preferred shareholders; and
•required shareholders to enter into a 180-day lock-up period upon an initial public offering.
The Stockholders Agreement terminated automatically upon completion of the IPO.
Eighth Amended and Restated Investor Rights Agreement
In connection with the Series D Convertible Preferred Stock financing in March 2015, we entered into the Eighth Amended and Restated Investor Rights Agreement (the "Investor Rights Agreement") with certain of our investors, including the Longitude Funds, the Boston Millennia Funds, Frazier Healthcare VI, L.P., Skyline Venture Partners V, L.P., TPG Biotechnology Partners IV, L.P. and RA Capital Healthcare Fund, LP The Investor Rights Agreement, among other things:
•imposes restrictions on the transfer of capital stock;
•grants holders of our outstanding convertible preferred stock certain registration rights following an initial public offering;
•grants holders of our outstanding convertible preferred stock certain pre-emptive rights with respect to certain issuances of our securities;
•imposes certain affirmative and negative covenants on us, including an obligation for us to deliver periodic financial statements and budgets to any holder of at least 1 million shares of our preferred stock;
•grants board observer rights to the Longitude Funds, Skyline Venture Partners V, L.P., Boston Millennia Partners and TPG Biotechnology Partners IV, L.P. so long as such investors hold at least 2 million shares of our preferred stock;
•requires us to prepare and submit to the SEC a registration statement on Form S-1 for an initial public offering of our common stock and to use our best efforts to cause the registration statement to be declared effective; and
•requires shareholders to enter into a 180-day lock-up period upon an initial public offering.
The provisions in the Investor Rights Agreement granting certain pre-emptive rights terminated automatically upon closing of the IPO.
Management Rights Letters
We entered into management rights letters with certain of our shareholders, including the Boston Millennia Funds, the Longitude Funds, Frazier Healthcare VI, L.P., Skyline Venture Partners V, L.P. and Aperture Venture Partners III, L.P. The management rights letters granted certain management rights in the event that such shareholder is not represented on our Board, as well as certain inspection rights. The management rights letters terminated automatically upon completion of the IPO.
Table of Contents
Employment Agreements
We have entered into employment agreements with certain of our named executive officers that provide for salary, bonuscash incentives, and severance compensation. For more information regarding these employment agreements, see "Executive“Executive Compensation—Employment Agreements"Agreements” and "Executive“Executive Compensation—Potential Payments Upon a Termination or Change of Control."”
Equity Issued to Executive Officers and Directors
We have granted common stock and/or stock options to our named executive officers and non-employee directors, as more fully described in "Executive“Executive Compensation—Employment Agreements," "Executive” “Executive Compensation—Equity Awards During Fiscal Years EndedYear ended December 31, 20152020” and 2014" and "Proposal“Proposal 1: Election of Directors—Corporate Governance—Compensation of Non-Employee Directors."”